MYOPIA / PRESBYOPIA MODELS
Carefully Focused.
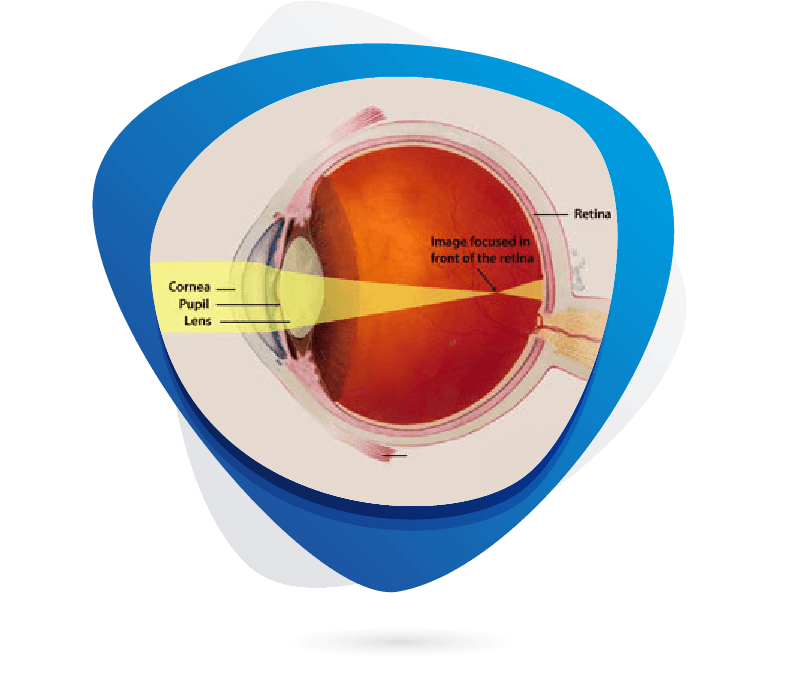
Myopia, or nearsightedness, is a growing global public health concern, with prevalence projected to affect nearly 50% of the world’s population by 2050. The therapeutic market is expanding rapidly, with estimates suggesting it could exceed $25 billion by the early 2030s, driven by increasing incidence in children and adolescents. Current treatments focus on slowing progression, including low-dose atropine eye drops, orthokeratology lenses, multifocal contact lenses, and lifestyle modifications like increased outdoor time. Recent R&D efforts target underlying molecular mechanisms such as scleral remodeling and retinal signaling; key targets include dopamine pathways, muscarinic acetylcholine receptors (notably M1 and M4), and adenosine receptors. Preclinical studies commonly use animal models like guinea pigs, chicks, and tree shrews to investigate axial elongation and test anti-myopia agents. Novel therapeutics under investigation include selective muscarinic antagonists, dopamine agonists, and gene or light-based interventions aimed at regulating eye growth.
MODELS / SERVICES
Where pharmacology meets excellence:
- AAALAC and CNAS guidelines-adherent animal studies
- 24/7 access to PharmaLegacy representatives
- Completed over 400 FDA / CFDA IND filings
- FDA Part 11 compliant
Over half of the top 20 pharmaceutical companies can’t be wrong. Choose PharmaLegacy.
Top Pharmaceutical Companies Trust PharmaLegacy For Their Preclinical Ophthalmology Studies
Your preclinical ophthalmology studies call for flawless and affordable ophthalmology models. Download our Ophthalmology brochure and see how our models and decades of expertise can give you the clarity you need in your pipeline.

We’re ready when you are.
Tell us your pharmacology challenges & start your study in as little as 2 weeks!

