Retinal Vein Occlusion
Unclog Your Pipelines with RVO Models.
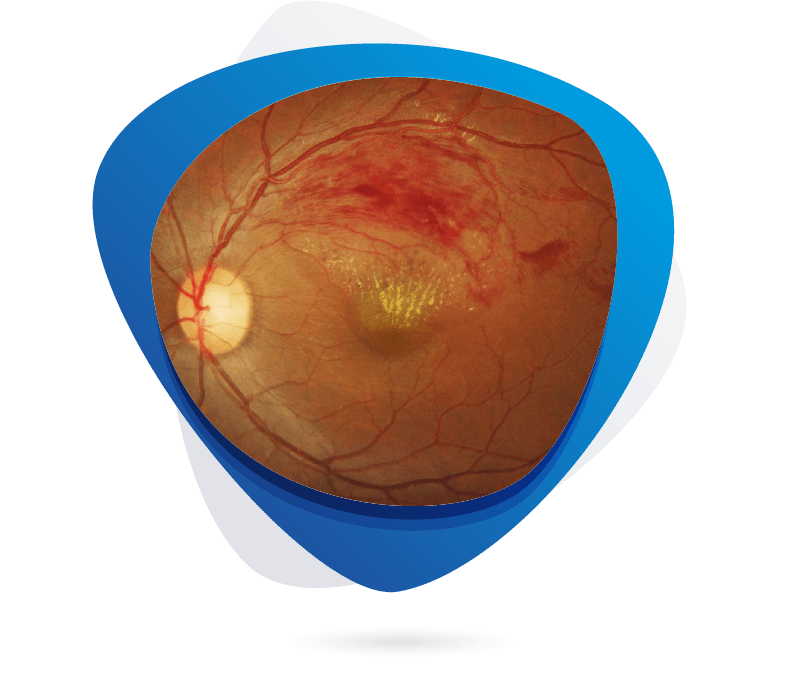
Retinal Vein Occlusion (RVO) is a common retinal vascular disorder characterized by the blockage of retinal veins, leading to vision loss due to macular edema, retinal ischemia, and neovascular complications. Clinically, RVO is classified into branch (BRVO) and central (CRVO) types, both of which significantly impair quality of life, especially in older adults with comorbidities like hypertension or diabetes. Current treatments primarily involve intravitreal anti-VEGF agents and corticosteroids to manage macular edema; however, these require frequent injections and show variable efficacy, particularly in ischemic cases. Unmet medical needs include effective therapies for non-responders, neuroprotection, and agents that address the underlying vascular pathology. Emerging drug targets under investigation include angiopoietin/Tie2 signaling, inflammatory cytokines (e.g., IL-6, TNF-α), and molecules involved in thrombosis and vascular stability.
MODELS / SERVICES
Where pharmacology meets excellence:
- AAALAC and CNAS guidelines-adherent animal studies
- 24/7 access to PharmaLegacy representatives
- Completed over 400 FDA / CFDA IND filings
- FDA Part 11 compliant
Over half of the top 20 pharmaceutical companies can’t be wrong. Choose PharmaLegacy.
Top Pharmaceutical Companies Trust PharmaLegacy For Their Preclinical Ophthalmology Studies
Your preclinical ophthalmology studies call for flawless and affordable ophthalmology models. Download our Ophthalmology brochure and see how our models and decades of expertise can give you the clarity you need in your pipeline.

We’re ready when you are.
Tell us your pharmacology challenges & start your RVO study in as little as 2 weeks!

