PTERYGIUM MODELS
Carefully designed Pterygium Models.
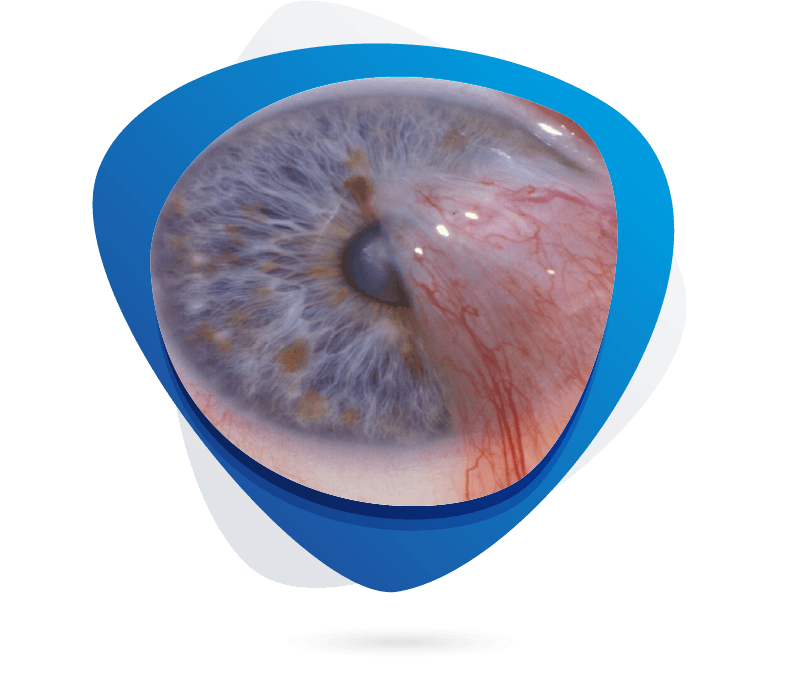
Pterygium is a common fibrovascular growth of the conjunctiva onto the cornea, with an estimated global prevalence of about 12% (95% CI 11–14%) that increases with age and UV exposure. Current management begins with lubricating eye drops and topical corticosteroids for mild cases, advancing to surgical excision with conjunctival autograft or amniotic membrane transplantation—often augmented by mitomycin C or beta‑radiation to reduce recurrence—and, increasingly, off‑label anti‑VEGF injections for recurrent disease. Recent R&D has focused on antifibrotic and targeted molecular therapies—such as pirfenidone down‑regulating TGF‑β and MMP‑1 in human pterygium fibroblasts and small‑molecule inhibitors of MMP‑14—offering promise for pharmacologic recurrence prevention. Key pathogenic targets under investigation include MMP‑14 (and related ADAM9/10/17), PDGFR‑β, VEGF, TGF‑β and other matrix‑remodeling pathways, while preclinical models in rabbits (subconjunctival NIH 3T3/Matrigel or UV‑induced lesions) and mice (human epithelial cell xenografts) now faithfully recapitulate pterygium‑like lesions for testing novel therapies.
MODELS / SERVICES
Where pharmacology meets excellence:
- AAALAC and CNAS guidelines-adherent animal studies
- 24/7 access to PharmaLegacy representatives
- Completed over 400 FDA / CFDA IND filings
- FDA Part 11 compliant
Over half of the top 20 pharmaceutical companies can’t be wrong. Choose PharmaLegacy.
Top Pharmaceutical Companies Trust PharmaLegacy For Their Preclinical Ophthalmology Studies
Your preclinical ophthalmology studies call for flawless and affordable ophthalmology models. Download our Ophthalmology brochure and see how our models and decades of expertise can give you the clarity you need in your pipeline.

We’re ready when you are.
Tell us your pharmacology challenges & start your study in as little as 2 weeks!

